More Information
Submitted: February 19, 2026 | Approved: February 25, 2026 | Published: February 26, 2026
How to cite this article: Thamizhoviya G, Muthuvel A. Purgation Therapy in Neurological Disorders: Bridging Siddha Medicine and Modern Neuroscience. J Neurosci Neurol Disord. 2026; 10(1): 001-009. Available from:
https://dx.doi.org/10.29328/journal.jnnd.1001114
DOI: 10.29328/journal.jnnd.1001114
Copyright License: © 2026 Thamizhoviya G, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Neurological disorders; Siddha medicine; Purgation therapy; Gut–brain axis; Constipation; Neuroinflammation
Purgation Therapy in Neurological Disorders: Bridging Siddha Medicine and Modern Neuroscience
Ashley S Membreno Lopez* and Arumugam Muthuve
and Arumugam Muthuve
Department of Integrative Health and Translational Research, National Institute of Siddha, Ministry of Ayush, Govt of India, Affiliated to TN. Dr MGR Medical University, Tambaram Sanatorium, Chennai 600047, India
*Address for Correspondence: Dr. Gangadharan Thamizhoviya, Assistant Professor, Department of Integrative Health and Translational Research, National Institute of Siddha, Ministry of Ayush, Govt of India, Affiliated to TN. Dr MGR Medical University, Tambaram Sanatorium, Chennai 600047, India, Email: [email protected]
Neurological disorders such as Parkinson’s disease, multiple sclerosis, and functional neurological syndromes represent a growing global health burden. Increasing evidence indicates that chronic constipation is not merely a secondary symptom but an early and contributory factor associated with gut–brain axis dysregulation, systemic inflammation, and neuroimmune alterations. The Siddha system of medicine, a traditional medical system practised in India, has long emphasized purgation therapy (Virechanam) as a regulatory intervention for neurological dysfunctions attributed to imbalances in Vatham, the functional principle governing movement and neural activity. This narrative review examines the therapeutic relevance of Siddha-based purgation therapy in the context of contemporary neurogastroenterological and neuroscientific research. By synthesizing evidence from studies on gut dysbiosis, vagal signalling, immune modulation, and neuroinflammation, the review explores conceptual convergences between traditional Siddha principles and modern understandings of the gut–brain axis. Chronic constipation, recognized both as a traditional marker of systemic imbalance and a prodromal feature of neurodegenerative disease, serves as a central point of intersection. Rather than asserting clinical equivalence or therapeutic substitution, this review highlights biological plausibility, shared mechanistic pathways, and translational opportunities through which gut-centred interventions, including purgation-based strategies, may complement contemporary neurological care. The findings support the need for further standardized clinical and mechanistic studies to evaluate the role of traditional detoxification practices within integrative approaches to neurological disorders.
Neurological disorders such as Parkinson’s disease, multiple sclerosis, epilepsy, and functional neurological syndromes have emerged as significant global health burdens, contributing to disability-adjusted life years (DALYs) and reduced quality of life [1,2]. Despite advancements in neuroimaging, diagnostics, and pharmacotherapy, modern neurology faces several key challenges. These include the lack of effective disease-modifying therapies, particularly for neurodegenerative disorders like Parkinson’s disease and multiple sclerosis, as well as the incomplete understanding of disease pathogenesis and the complex, multifactorial nature of these conditions [3]. Furthermore, modern treatments often overlook the gastrointestinal dysfunctions associated with neurological diseases, such as chronic constipation, which have increasingly been recognized as contributors to systemic inflammation, neuroinflammation, and immune dysregulation [4,5]. In contrast, the traditional Indian medical system, especially Siddha medicine, had approached the neurological dysfunctions through a holistic, systems-oriented perspective. It attributed many of these conditions to imbalances in Vatham, the fundamental humour responsible for movement and neural regulation [6,7].
The Siddha principle of “Viresanathal Vatham Thazhum”, literally meaning “purgation pacifies aggravated Vatham, emphasizes purgation therapy (Virechanam) as a regulatory intervention that extends beyond bowel evacuation to the correction of systemic imbalance [8,9]. Within Siddha medicine, purgation is traditionally understood as a preparatory and restorative process intended to normalize internal functional derangements and enhance the body’s responsiveness to subsequent therapies. In contrast, modern neurological care has historically placed limited emphasis on gastrointestinal cleansing or detoxification as part of disease management, revealing a conceptual gap that warrants further scientific exploration [9,10].
Purgation therapy has traditionally been employed in Siddha medicine as a gut-centred regulatory intervention aimed at addressing systemic imbalances associated with neurological dysfunction. Although its mechanisms were historically framed within humoral theory, emerging insights from gut–brain axis research offer a contemporary biological context in which such practices may be examined. Accordingly, the present narrative review explores the intersections between Siddha purgation therapy (Virechanam) and modern neurogastroenterological perspectives, with particular emphasis on chronic constipation, gut–brain communication, and neuroinflammation in neurological disorders.
This manuscript is a narrative, concept-driven review aimed at synthesizing traditional Siddha perspectives on purgation therapy with contemporary evidence on gut–brain axis mechanisms. Relevant peer-reviewed literature was identified through non-systematic searches of PubMed, Scopus, and Google Scholar using combinations of keywords including “Siddha medicine,” “Virechanam,” “gut–brain axis,” “constipation,” “neuroinflammation,” and “neurodegeneration.” The objective was not to perform a systematic meta-analysis but to explore conceptual convergence and biological plausibility across traditional and biomedical frameworks. As this work does not involve primary data collection or human participants, ethical approval and participant recruitment were not applicable.
Neurological disorders- siddha perspective
In Siddha philosophy, the three body humours Vatham, Pitham, and Kapham govern the body’s functions, with Vatham playing a critical role in maintaining neurological and motor functions. Characterized by the qualities of dryness, lightness, coldness, roughness, and mobility, Vatham regulates both physical and neurological movements, particularly with a dominant presence below the navel [11]. Vatham is classified into ten subtypes (Dasavatham), each responsible for specific physiological functions. For neurological dysfunctions, the most relevant subtypes are:
- Piranan: Regulation of respiration and circulation.
- Viyanan: Coordination of body movements and neurological functions.
- Samanan: Digestion and assimilation, impacting brain-gut interactions.
- Naagan: Cognition and mental faculties.
- Udhanan: Speech, which is linked to neurological control.
- Devathathan: Emotional expressions such as anger, which can affect neurological balance.
Disturbances in these subtypes can result in a range of neurological dysfunctions, and purgation therapy is traditionally used to restore balance in these humours by eliminating excess Vatham and addressing neurological and motor impairments [11,12].
Classical Siddha texts described several causes for Vatham derangement, including dietary irregularities (such as consumption of dry, cold, or light foods), environmental exposure (cold winds, seasonal changes), emotional stress, ageing, and chronic illness [13]. The resulting symptoms were reported as tremors, stiffness, numbness, joint pain, mental confusion, insomnia, and impaired locomotion, many of which closely resembled the presentations of modern neurological disorders [14,15]. The pathogenesis of Vatham imbalance originated in the gastrointestinal tract, particularly the colon, before spreading systemically and localizing in vulnerable regions like the nervous system and joints [9].
Classical Siddha texts such as Agathiyar Gunavakadam, Theraiyar Yemaga Venba, and Yugi Vaithiya Chinthamani described Vatham derangement as a root cause of both neurological and behavioral disturbances. “Chronic constipation”, in particular, was viewed not merely as a symptom but as a systemic disruptor leading to the accumulation of toxins and the stagnation of life energy (vayu). These pathophysiological insights showed significant alignment with modern observations of neurodegenerative disorders, including Parkinson’s disease, multiple sclerosis, and certain dementias, where systemic inflammation and impaired gut function are now recognized as contributing factors [16,17]. Although founded on different paradigms, modern neurology reflected many of these ancient principles. Clinical features such as tremors, rigidity, gait disturbances in Parkinson’s disease, and episodic demyelination in multiple sclerosis closely reflected Siddha’s descriptions of Vatha imbalance [13,18-21]. This convergence of traditional diagnostics with modern pathology highlighted the therapeutic potential of Vatham-pacifying strategies, particularly purgation therapy (Virechanam) to restore the systemic balance and address the gut–brain axis dysfunctions commonly seen in these disorders.
Purgation therapy: Siddha principles and procedures
Purgation therapy was one of the crucial purification therapies in Siddha medicine, specifically used in expelling the aggravated Vatham from the gastrointestinal tract and reducing the Vatham-predominant diseases. Siddha texts prescribed purgation therapy for a spectrum of Vatha-predominant disorders, including chronic constipation, hemiplegia, facial palsy, epilepsy, neuralgia, tremors, insomnia, and mental disturbances. These conditions were attributed to the derangement of Vatham within the intestinal and systemic pathways [22,23]. Unlike mere bowel cleansing, the purgation therapy was conceived as a systemic detoxification procedure that influenced not only the digestive organs but also the nervous system, and immune pathways, particularly via the gut–brain axis [9,24]. Siddha-based principle of “Viresanathal Vatham Thazhum” exhibited reduction of the aggravated Vatham through purgation therapy; this practice could reflect Siddha’s integrated approach to modern neurological care via gastrointestinal detoxification [9,25]. This resetting of the internal milieu could be considered essential in restoring the functional homeostasis.
Purgation therapy was restricted to seasonal specificity, for instance, Ricinus communis (castor oil) given in summer and Croton tiglium (Nervalam) in rainy and winter seasons. Terminalia chebula (Kadukkai), considered Siddha’s key Vatham-reducing herb, which was valued as a digestive stimulant, detoxifier, nervine tonic, and gut-channel cleanser, now supported by pharmacological studies reporting its antioxidant, neuroprotective, anti-inflammatory, and gut microbiota-modulating effects, making it a potential agent in neuroinflammatory and neurodegenerative contexts [26,27]. Post-purgation, patients were monitored for vital signs and therapeutic outcomes and were advised on a regulated bland diet (Kanji), hydration, and rest to restore normalcy [8,9,24,28]. This seasonal and systematically monitored purgation framework in Siddha paralleled modern neuroscience insights, where chronic constipation and gut dysbiosis have been increasingly recognized as early markers and mediators of gut–brain axis dysfunction leading to neurodegeneration.
Constipation in modern neuroscience: Gut–brain axis as a pathway to neurodegeneration
Constipation was one of the most frequent non-motor symptoms of Parkinson’s disease (PD), affecting 61.4% of patients, with 24.5% experiencing it before the motor symptoms appeared. This symptom was linked to more severe motor and non-motor issues, including older age, longer disease duration, and poorer quality of life [29]. Constipation not only impacted the daily functioning but also served as a prodromal marker for neurodegeneration. In recent years, modern neuroscience has increasingly recognized the gut–brain axis (GBA) as a complex, bidirectional communication network involving the enteric nervous system (ENS), central nervous system (CNS), autonomic signalling, neuroendocrine regulation, immune modulation, and the intestinal microbiota [30,31]. Within this framework, the gut microbial dysbiosis, systemic inflammation, and ENS dysfunction have been implicated in a wide range of neurodegenerative disorders, including Parkinson’s disease, Alzheimer’s disease (AD), and other neuropsychiatric conditions [30,32-35].
Chronic constipation was recognized as a key pathological trigger of gut–brain axis disruption, leading to bowel stagnation, depletion of beneficial microbes such as Bifidobacterium and Lactobacillus, and an increase in pro-inflammatory taxa. This dysbiosis altered the short-chain fatty acid (SCFA) profiles, elevated the intestinal permeability, and promoted low-grade systemic inflammation [36,37]. Further, the microbial endotoxins such as lipopolysaccharides (LPS) translocated into the systemic circulation, which crossed the blood–brain barrier (BBB), activated the microglia, and contributed to neuroinflammatory cascades [38,39]. The key inflammatory mediators, especially interleukin-6 (IL-6), were found to be elevated in both the peripheral blood and cerebrospinal fluid of patients with PD, AD, and multiple sclerosis [40-42]. This linked chronic constipation to systemic inflammation and neurodegeneration, highlighting how microbial metabolites and inflammatory mediators exerted their effects not only via neural pathways but also through humoral (systemic circulatory and immune) routes within the gut–brain axis [43,44]. Supporting this, salivary cytokine levels were shown to correlate with the gut inflammation in cerebral palsy patients [45], while germ-free animal models also further confirmed ENS dysfunction and microbiota imbalance as central to neurodegenerative symptomatology [43,46].
Recent research on constipation in Parkinson’s disease highlighted its significant impact on patients’ well-being, but effective treatments remained scarce. While diet interventions with probiotics, lubiprostone, and botulinum toxin showed some promise, further research was needed to establish stronger recommendations [47]. This was particularly relevant as recent literature suggested that intestinal inflammation contributed to PD’s pathogenesis, linking environmental factors like altered gut microbiota to neurodegeneration in genetically susceptible patients [48]. The overlap between PD and neuroimmune interference by the gut-brain axis signalling could significantly provide opportunities to develop new treatment strategies, potentially improving the outcomes for those with constipation-related symptoms.
Vagal signaling and the neuroimmune interface
The vagus nerve played a pivotal role in gastrointestinal regulation. Afferent fibres relayed via mechanical and microbial stimuli from the gut to the brainstem, while the efferent signals influenced the gut motility, secretion, and immune homeostasis [49,50]. In chronic constipation, the vagal hypoactivity impaired the neuroendocrine feedback loops, which significantly reduced the anti-inflammatory regulation and facilitated the central immune activation [51]. Among the vagal-mediated factors, butyrate, a key SCFA, was shown to regulate the microglial function and preserve the intestinal barrier integrity. Its depletion was further linked to compromising the blood-brain barrier and increased neuroinflammatory pathway [49,52,53]. Based on these insights, connecting the gut-brain axis via the vagus nerve could be regulated by factors including genetic diversity and environmental conditions among patients with constipation and neurodegenerative conditions.
Genetic, environmental, and epidemiological evidence
Emerging evidence also pointed to genetic susceptibilities linking gut inflammation and neurodegeneration. Mutations in genes such as NOD2 and TREM2, involved in intestinal barrier regulation and immune signalling, were observed in PD and AD patients [54,55]. Environmental factors like antibiotic overuse, low-fibre diets, and sedentary behavior further aggravated the microbial dysbiosis and constipation [56]. Many large-scale clinical and epidemiological studies corroborated these findings. The UK Biobank cohort involving over 500,000 individuals reported increased risk of cognitive impairment and dementia in constipated individuals [57]. Similarly, an 8-year longitudinal study [58] from the Parkinson’s Incidence Cohorts Collaboration revealed that early and severe constipation was linked to faster progression to dementia in PD patients. A meta-analysis by Yao, et al. [59] involving over 3 million individuals reported a pooled odds ratio (OR) of 2.36 for PD among those with constipation, highlighting it as a prodromal symptom in 20% of PD patients versus 11% in controls. Moreover, Warnecke, et al. [60] linked enteric α-synuclein accumulation to gut motility issues, further supporting the gut-origin hypothesis. About AD, a South Korea–Japan bi-national cohort [61] found hazard ratios of 2.04 and 2.82 for constipation-associated AD development. In addition to this, animal models also supported these findings, where loperamide-induced gut dysmotility in AD mice accelerated the amyloid-β accumulation and microglial activation, while Tg2576 and A53T transgenic models showed that GI dysfunction preceded the CNS pathology [4,46,62]. Therefore, based on these supporting evidences, it could be ascertained that the external and genetic factors played an important role in gut-brain axis regulation, which further necessitated the early markers and therapeutic strategy for the dysbiosis.
From early markers to therapeutic targets
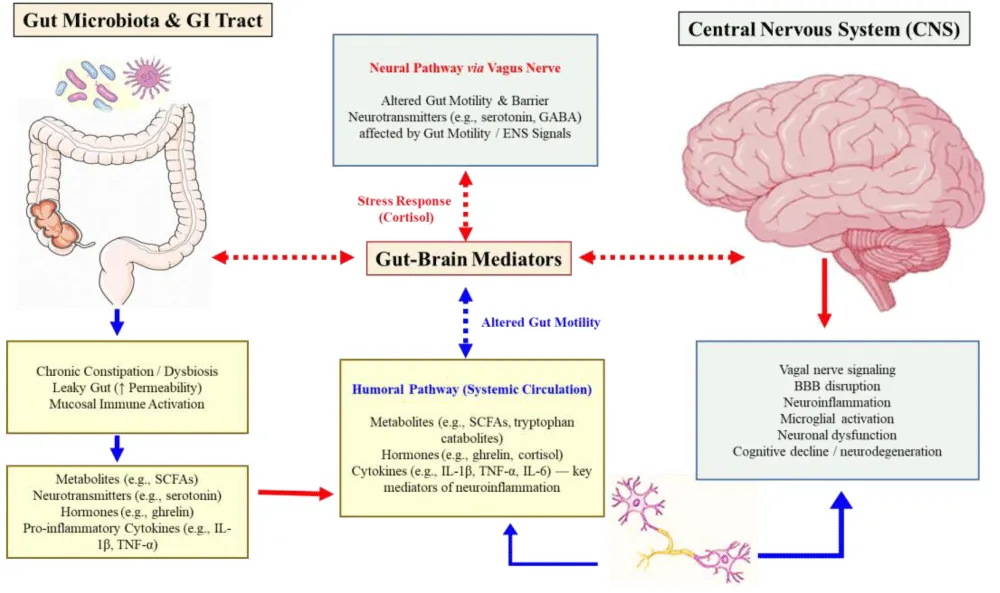
Constipation was also associated with prodromal features of Lewy body diseases, including REM sleep behavior disorder and neurological dysfunction [63,64]. Functional MRI studies correlated constipation severity with insula and orbitofrontal cortex disruptions in PD [65,66]. A meta-analysis by Wang & Jiang [67] reported an odds ratio (OR) of 1.33 for cognitive impairment in PD patients with constipation. Mechanistically, α-synuclein propagation via enteroendocrine cells and vagal routes was proposed [33], indicating the gut as a starting point of neuropathology. Figure 1 illustrates the bidirectional signalling across neural and humoral pathways, emphasizing afferent vagal signaling, HPA axis feedback, systemic inflammation, and gut barrier modulation.
Figure 1: Illustrated the bidirectional communication between the gut microbiota and the central nervous system through neural and humoral pathways, where the dotted red arrow represented the primary vagal neural connection between the gut and the brain, the solid red arrow indicated the stress-related feedback loop involving the hypothalamic–pituitary–adrenal (HPA) axis and cortisol, the solid blue arrow denoted the humoral transmission of microbial and host-derived mediators such as metabolites, cytokines, and hormones to the central nervous system via systemic circulation, and the dotted blue arrow depicted modulatory feedback mechanisms, such as changes in gut motility or intestinal permeability that influenced brain signaling [43,44].
Gut microbiota–CNS axis in constipation-induced neurodegeneration
Recent investigations in modern neuroscience regarding the relationship between chronic constipation and gut dysbiosis closely resonate with the Siddha medical system, in which the intestine (Kudal) is traditionally regarded as the seat of Vatham—the functional principle governing movement, neurological stability, and systemic balance [68]. In Siddha medicine, constipation is viewed as an early manifestation of Vatha derangement, leading to impairments in neuromuscular coordination and cognitive clarity. This conceptual overlap provides a relevant framework for integrative therapeutic exploration. Modern neuroscience emphasizes the role of gut microbiota–CNS signalling, neuroinflammation, and vagal nerve dysfunction in the pathophysiology of neurodegenerative disorders, while Siddha medicine focuses on gut-centred detoxification and humoral rebalancing. Traditional purgative therapy is intended to relieve colonic stagnation and restore Vatham equilibrium [27], and may conceptually align with contemporary neurobiological strategies targeting gut–brain axis dysfunction.
Taken together, these perspectives position the gut as a critical upstream regulator of neurological health. Constipation, once considered a secondary symptom, has emerged as a modifiable risk marker and early indicator of neurodegenerative vulnerability. Integrating Siddha-based gastrointestinal approaches with modern gut–brain axis–modulating strategies may therefore offer a complementary framework for addressing neurodegenerative processes, warranting further translational and clinical investigation.
Principles of purgative therapy in integrating siddha and modern neuroscience via gut–brain axis
Building upon the shared recognition of the gut as central to neurological regulation, the integration of purgative therapy with modern neuroscience provided a compelling framework for addressing the gut–brain axis. In Siddha medicine, purgation therapy was regarded as a primary therapeutic step, aimed at eliminating stagnation and rebalancing the body’s internal milieu before any further treatment was initiated. Modern neuroscience, through the study of the gut microbiota, similarly emphasized that bowel dysfunction contributed to systemic inflammation, impaired barrier integrity, and dysregulated neuroimmune signalling.
Constipation, long dismissed as trivial, had in fact emerged as an early biomarker and modifiable risk factor for Parkinson’s disease, Alzheimer’s disease, and other neurodegenerative conditions [69,70]. Experimental studies demonstrated that constipation-induced dysbiosis disrupted the gut and blood–brain barriers, activated microglia, and promoted neuroinflammation [5,71]. Siddha texts anticipated this association, regarding constipation as a manifestation of Vatha derangement that impaired neuromuscular coordination and mental clarity. Thus, both systems converged on the view that bowel dysfunction represented an upstream driver of neurological decline. This conceptual overlap was further illustrated in Table 1, which presented a comparative view of Siddha and modern biomedical perspectives on the gut–brain relationship, particularly in the context of constipation-induced dysbiosis and neuroinflammation. While Siddha medicine emphasized vitiated Vatham as the cause of neurological imbalance, modern medicine focused on gut barrier disruption, cytokine elevation, and microbiota dysregulation. Despite differing terminologies, both systems recognized bowel dysfunction as a root cause of neurological deterioration.
| Table 1: Comparison of Siddha and modern medical perspectives on the gut–brain connection, highlighting core concepts, causes, communication pathways, symptoms, and treatment approaches adopted from [6,9,43,74]. | ||
| Aspect | Siddha medicine | Modern medicine |
| Core Concept | Vatham, which governs the nervous system, originates in the gut | The Enteric Nervous System (ENS), referred to as the "second brain," is located in the gut |
| Cause of Dysfunction | Indigestion, constipation, and toxin buildup disturb Vatham, which disrupts brain functions. | Gut dysbiosis, inflammation, and intestinal barrier dysfunction affect the brain via the vagus nerve and immune pathways. |
| Communication Pathway | Vitiated Vatham affects the body and mind, leading to dysfunction in mental and neurological functions. | Gut–Brain Axis: Bidirectional signalling via the vagus nerve, hormones, and cytokines |
| Symptoms | Tremors, confusion, seizures, mental dullness (due to Vatham imbalance originating in the gut) | Depression, anxiety, cognitive decline, Parkinson’s (linked to gut imbalances) |
| Treatment Focus | Purgation (Virechanam), gut detox, and Vatham-pacifying herbs | Addressing gut microbiome imbalances and intestinal inflammation via appropriate medical treatments |
| Preventive View | Daily bowel movement is crucial for maintaining nervous system balance | Maintaining gut health through a balanced diet and lifestyle supports overall brain function |
Gut–brain connection: Siddha vs. modern medicine
Importantly, the role of purgation extended beyond mere symptom relief. Modern studies demonstrated that bowel cleansing induced profound yet beneficial shifts in the gut microbiota. Polyethene glycol–based cleansing had transiently reduced microbial load, which was subsequently followed by the restoration of diversity within two weeks, including enrichment of beneficial taxa such as Bifidobacterium and Lactobacillus [72,73]. Subsequent work confirmed that bowel cleansing altered microbial metabolites, including short-chain fatty acids (SCFAs), which played a crucial role in neuroimmune and epithelial homeostasis [52,74].
This sequence from transient microbial depletion, to restoration and enrichment of SCFA-producing taxa, to downstream immunomodulation and neuroprotective effects demonstrated a cascade in which purgation influenced the gut–brain axis at multiple levels. Beyond microbial changes, purgative interventions were also found to modulate host immune responses. Reduced levels of pro-inflammatory cytokines such as IL-6 and TNF-α and increased regulatory T-cell activity were observed following colon cleansing, suggesting systemic immunomodulation [75-77]. Improvements in gut barrier integrity were documented, with tighter junction expression and decreased intestinal permeability, thereby lowering translocation of endotoxins like LPS into the circulation [78,79]. These systemic effects extended to the central nervous system. Clinical studies linked bowel cleansing to improvements in mood, anxiety, fatigue, and sleep quality [33,67,80-85]. Such findings resonated with the Siddha principle that systemic cleansing not only restores somatic balance but also prepares the body and mind for effective therapeutic response.
This conceptual overlap highlighted a critical therapeutic gap. While modern medicine often prioritises symptomatic management of neurological disorders, Siddha medicine emphasized preparatory cleansing as the essential first step. The integration of purgative therapy into contemporary neurology should therefore be considered indispensable for enhancing gut–brain axis modulation, reducing systemic inflammation, and improving the efficacy of subsequent pharmacological and rehabilitative interventions. Although purgation therapy demonstrated conceptual and physiological relevance, its integration into modern neurology faced several challenges. Standardized protocols for timing, frequency, and method were lacking, and large-scale clinical trials or mechanistic studies remained scarce. Regulatory frameworks and conventional clinical guidelines did not formally recognize purgation, limiting its adoption. Nonetheless, structured purgation holds promise as a preventive and therapeutic approach by modulating gut–brain interactions, systemic inflammation, and neuroimmune pathways. Future research with standardized procedures and translational studies could validate its efficacy, bridging traditional wisdom with contemporary neurological care.
Scope and limitations
This review is conceptual and integrative in nature and does not claim direct clinical efficacy of purgation therapy for neurological disorders. While mechanistic parallels between Siddha purgation practices and modern gut–brain axis research are highlighted, differences in formulations, dosing, and procedural contexts must be acknowledged. Much of the supporting evidence is derived from associative clinical studies, experimental models, and indirect biological analogies. Consequently, well-designed clinical trials, standardized protocols, and mechanistic investigations are required before purgation therapy can be evaluated within evidence-based neurological practice.
Purgation therapy has long been regarded in Siddha medicine as a foundational approach to correcting systemic imbalance, particularly in conditions attributed to Vatham derangement. When viewed through contemporary scientific frameworks, this traditional practice aligns with emerging evidence on the gut–brain axis, neuroimmune signalling, and microbiota-driven modulation of neurological function. Rather than representing a disease-specific intervention, purgation may be conceptualized as a gut-centred adjunctive strategy aimed at addressing chronic constipation, intestinal dysbiosis, and low-grade systemic inflammation mechanisms increasingly implicated in neurodegenerative and functional neurological disorders. This review does not advocate substitution of conventional neurological care but highlights the biological plausibility of integrating gut-focused strategies within preventive and complementary frameworks.
To advance this field, well-designed pilot clinical trials evaluating standardized Siddha purgative formulations such as Terminalia chebula–based preparations or regulated castor oil protocols in constipation-associated neurological disorders are warranted. Mechanistic studies assessing microbiota composition, short-chain fatty acids, inflammatory markers, and neuroimaging correlates, alongside preclinical investigations in transgenic models, would help clarify potential effects on α-synuclein aggregation, microglial activation, and barrier integrity. Establishing standardized dosing, safety parameters, and microbiome endpoints will be critical for translating traditional purgation practices into rigorously evaluated integrative neurological strategies.
Declarations
Acknowledgement: We sincerely thank the Department of Integrative Health and Translational Research for their encouragement and the National Institute of Siddha for their valuable support.
Ethical statement: This review paper has been prepared in accordance with the ethical standards expected in academic research and publication. The authors confirm that all the sources have been properly cited and acknowledged, and there is no fabrication, falsification or manipulation of information.
Informed consent: This work does not involve any new human or animal research data. Instead, it synthesizes and analyzes previously published studies, respecting the intellectual property rights and academic integrity.
Conflicts of interest: The authors declare no competing interests. The authors declare that the manuscript has not been submitted or published elsewhere for publication.
- Thakur KT, Albanese E, Giannakopoulos P. Neurological disorders. In: Patel V, Chisholm D, Dua T, Laxminarayan R, Medina-Mora ME, editors. Mental, neurological, and substance use disorders: disease control priorities. 3rd ed. Vol. 4. Washington (DC): The International Bank for Reconstruction and Development / The World Bank; 2016 Mar 14. Chapter 5. Available from: https://www.ncbi.nlm.nih.gov/books/NBK361945/
- Feigin VL, Vos T, Nichols E, Owolabi MO, Carroll WM, Dichgans M, et al. The global burden of neurological disorders: translating evidence into policy. Lancet Neurol. 2020;19(3):255-265. Available from: https://doi.org/10.1016/s1474-4422(19)30411-9
- Gadhave DG, Sugandhi VV, Jha SK, Nangare SN, Gupta G, Singh SK, Paudel KR, et al. Neurodegenerative disorders: mechanisms of degeneration and therapeutic approaches with their clinical relevance. Ageing Res Rev. 2024;102357. Available from: https://doi.org/10.1016/j.arr.2024.102357
- Kim JE, Park JJ, Lee MR, Choi JY, Song BR, Park JW, Hwang DY, et al. Constipation in Tg2576 mice model for Alzheimer’s disease associated with dysregulation of mechanism involving the mAChR signaling pathway and ER stress response. PLoS One. 2019;14(4):e0215205. Available from: https://doi.org/10.1371/journal.pone.0215205
- Lin X, Liu Y, Ma L, Ma X, Shen L, Ma X, Chen X, et al. Constipation induced gut microbiota dysbiosis exacerbates experimental autoimmune encephalomyelitis in C57BL/6 mice. J Transl Med. 2021;19:1-16. Available from: https://doi.org/10.1186/s12967-021-02995-z
- Shukla SS, Saraf S. Fundamental aspect and basic concept of siddha medicines. Syst Rev Pharm. 2011;2(1). Available from: https://www.sysrevpharm.org/articles/fundamental-aspect-and-basic-concept-of-siddha-medicines.pdf
- Mahendiran M, Gowri V. Parallel analysis of Oruthalai Vaatham in Siddha literature with migrainous neuralgia. Available from: https://velumailusiddha.dharmagroups.in/parallel-analysis-of-oruthalai-vaatham-siddha-literature-with-migrainous-neuralgia/
- Rajeshwari M. Understanding the concept of purgation in Siddha medicine: a review. Int J Ayurveda Pharma Res. 2022;10(6). Available from: https://doi.org/10.47070/ijapr.v10i6.2324
- Thanigainathan A, Selvalenin C, Lakshmi Kantham T, Vetha Merlin Kumari H, Nalini Sofia H, Mary Sharmila C, Anbarasan B, Meenakumari R. Viresanam – purgation therapy in Siddha. J Siddha. 2021;5(1):52-60. Available from: https://jos.nischennai.org/index.php/jos/article/view/34
- Barik S, Prasad BS. Prescribing patterns of Virechana karma in terms of Shuddhi Lakshana. Indian J Health Sci Biomed Res KLEU. 2019;12(2):139-153. Available from: https://kleijhsbr.researchcommons.org/journal/vol12/iss2/9/
- Muthiah K, Ganesan K, Ponnaiah M, Parameswaran S. Concepts of body constitution in traditional Siddha texts: a literature review. J Ayurveda Integr Med. 2019;10(2):131-134. Available from: https://doi.org/10.1016/j.jaim.2019.04.002
- Chithra L, Christian GJ, Elansekaran S, Ramamoorthy M. An observational study to validate the symptomatology and to evolve the diagnostic methodology of Sirakkamba Vatham through Siddha diagnostic tools. Int J Ayurveda Pharma Res. 2020;8(2):31-37. Available from: https://ijapr.in/index.php/ijapr/article/view/1391/1086
- Council of Scientific and Industrial Research Traditional Knowledge Digital Library. Principles of Siddha [seasonal guidelines table]. Traditional Knowledge Digital Library. [cited 2025 Jun 30]. Available from: https://tkdl.res.in/tkdl/langdefault/Siddha/Sid_Principles.asp?GL=Eng
- Esakkimuthu S, Mutheeswaran S, Elankani P, Pandikumar P, Ignacimuthu S. Quantitative analysis of medicinal plants used to treat musculoskeletal ailments by non-institutionally trained Siddha practitioners of Virudhunagar district, Tamil Nadu, India. J Ayurveda Integr Med. 2021;12(1):58-64. Available from: https://doi.org/10.1016/j.jaim.2018.11.005
- Uma E, Mala T, Geetha AV, Priyanka D. A comprehensive survey of drug-target interaction analysis in allopathy and Siddha medicine. Artif Intell Med. 2024;102986. Available from: https://doi.org/10.1016/j.artmed.2024.102986
- Babu CS, Mahadevan M, Rao BS, Ranju V, Bipul R, Bhat A, Essa MM, et al. Management of Huntington's disease: perspectives from the Siddha system of medicine. In: Food for Huntington's disease. New York: Nova Science Publishers, Inc.; 2018;159-180. Available from: https://www.researchgate.net/publication/329865712_Management_of_Huntington's_disease_Perspectives_from_the_Siddha_system_of_medicine
- Gangadharan T, Arumugam M. Siddha medicine and modern neuroscience: a synergistic approach to neurological care. 3 Biotech. 2025;15(4):1-13. Available from: https://doi.org/10.1007/s13205-025-04265-x
- Sharma T, Meghwal CR, Prajapat M, Sharma AK. Conceptual study of Vata dosha with special reference to nervous system. J Ayurveda Integr Med Sci. 2024;9(2):134-140. Available from: https://jaims.in/jaims/article/view/3123
- Sowmya S, Sudha Revathy S, Meenakumari R. Therapeutic potential of Siddha herbomineral formulation “Kukkilathy Chorine” in Parkinsonism: a review. Int J Pharm Sci Rev Res. 2024;84(9):228-341. Available from: https://globalresearchonline.net/ijpsrr/v84-9/37.pdf
- Nisha J, Anbu N, Purushothaman I. A review on traditional herbs used in Parkinson’s disease (Nadukku Vatham). World J Pharm Res. 2017;7(16):631-637. Available from: https://doi.org/10.20959/wjpr20178-9217
- Mohanraj T. Vaadha noi thoguthi (Vaatha noi nidhanam–800). 1st ed. Chennai: ATSVS Siddha Medical College and Hospital; 2008;28.
- Uttarayan KA. Siddha maruthuvanga churukkam. 2nd ed. Chennai: Department of Indian Medicine and Homoeopathy. 2006;138-140, 654.
- Indian Medical Practitioners’ Co-operative Pharmacy and Stores Ltd. Formulary of Indian Medical Practitioners’ Co-operative Pharmacy and Stores Ltd. Chennai: IMPCOPS; 2018.
- Shanmughavelu HPIM. Noi naadal noi mudhal naadal thirattu. Part 1. Chennai: Department of Indian Medicine and Homoeopathy; 2007.
- Thayalini T, Prasad VM. Vatha suram (fever) in literatures of Siddha medicine. J. res. tradit. med. 2018;4(1):21-25. Available from: https://www.tmjournal.org/?mno=298118
- Guan Y, Tang G, Li L, Shu J, Zhao Y, Huang L, Tang J, et al. Herbal medicine and gut microbiota: exploring untapped therapeutic potential in neurodegenerative disease management. Arch Pharm Res. 2024;47(2):146-164. Available from: https://doi.org/10.1007/s12272-023-01484-9
- Sharma R, Kabra A, Rao MM, Prajapati PK. Herbal and holistic solutions for neurodegenerative and depressive disorders: leads from Ayurveda. Curr Pharm Des. 2018;24(22):2597-2608. Available from: https://doi.org/10.2174/1381612824666180821165741
- Suguna M, Arthi G, Mohanaprabha G, Dhineshraman G, Manjula V, Kavitha S, Krishnaveni M. An open-label non-randomized clinical trial on Siddha purgation therapy (Bhedhi) for pacifying Vatham disease using diagnostic tool Neikuri. Int J Ayurveda Pharma Res. 2022;10(11):95-102. Available from: https://doi.org/10.47070/ijapr.v10i11.2602
- Yu QJ, Yu SY, Zuo LJ, Lian TH, Hu Y, Wang RD, Zhang W, et al. Parkinson disease with constipation: clinical features and relevant factors. Sci Rep. 2018;8(1):567. Available from: https://www.nature.com/articles/s41598-017-16790-8b
- Foster JA, Rinaman L, Cryan JF. Stress and the gut-brain axis: regulation by the microbiome. Neurobiol Stress. 2017;7:124-136. Available from: https://doi.org/10.1016/j.ynstr.2017.03.001
- Xu J, Lu Y. The microbiota-gut-brain axis and central nervous system diseases: from mechanisms of pathogenesis to therapeutic strategies. Front Microbiol. 2025;16:1583562. Available from: https://doi.org/10.3389/fmicb.2025.1583562
- Chaudhry TS, Senapati SG, Gadam S, Mannam HPSS, Voruganti HV, Abbasi Z, Arunachalam SP, et al. The impact of microbiota on the gut–brain axis: examining the complex interplay and implications. J Clin Med. 2023;12(16):5231. Available from: https://doi.org/10.3390/jcm12165231
- Sampson TR, Tansey MG, West AB, Liddle RA. Lewy body diseases and the gut. Mol Neurodegener. 2025;20(1):14. Available from: https://doi.org/10.1186/s13024-025-00804-5
- Nakase T, Tatewaki Y, Thyreau B, Mutoh T, Tomita N, Yamamoto S, Taki Y, et al. Impact of constipation on progression of Alzheimer's disease: a retrospective study. CNS Neurosci Ther. 2022;28(12):1964-1973. Available from: https://doi.org/10.1111/cns.13940
- O’Riordan KJ, Moloney GM, Keane L, Clarke G, Cryan JF. The gut microbiota-immune-brain axis: therapeutic implications. Cell Rep Med. 2025;6(3). Available from: https://doi.org/10.1016/j.xcrm.2025.101982
- Clapp M, Aurora N, Herrera L, Bhatia M, Wilen E, Wakefield S. Gut microbiota’s effect on mental health: the gut-brain axis. Clin Pract (Lond). 2017;7(4):987. Available from: https://doi.org/10.4081/cp.2017.987
- Solanki R, Karande A, Ranganathan P. Emerging role of gut microbiota dysbiosis in neuroinflammation and neurodegeneration. Front Neurol. 2023;14:1149618. Available from: https://doi.org/10.3389/fneur.2023.1149618
- Su H, Mo J, Ni J, Ke H, Bao T, Xie J, Chen W, et al. Andrographolide exerts antihyperglycemic effect through strengthening intestinal barrier function and increasing microbial composition of Akkermansia muciniphila. Oxid Med Cell Longev. 2020;2020:6538930. Available from: https://doi.org/10.1155/2020/6538930
- Miri S, Yeo J, Abubaker S, Hammami R. Neuromicrobiology, an emerging neurometabolic facet of the gut microbiome? Front Microbiol. 2023;14:1098412. Available from: https://doi.org/10.3389/fmicb.2023.1098412
- Grebenciucova E, VanHaerents S. Interleukin 6: at the interface of human health and disease. Front Immunol. 2023;14:1255533. Available from: https://doi.org/10.3389/fimmu.2023.1255533
- Kummer KK, Zeidler M, Kalpachidou T, Kress M. Role of IL-6 in the regulation of neuronal development, survival and function. Cytokine. 2021;144:155582. Available from: https://doi.org/10.1016/j.cyto.2021.155582
- Chen W, Wang J, Yang H, Sun Y, Chen B, Liu Y, Zhan J, et al. Interleukin 22 and its association with neurodegenerative disease activity. Front Pharmacol. 2022;13:958022. Available from: https://doi.org/10.3389/fphar.2022.958022
- Carabotti M, Scirocco A, Maselli MA, Severi C. The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems. Ann Gastroenterol. 2015;28(2):203. Available from: https://pubmed.ncbi.nlm.nih.gov/25830558/
- Appleton J. The gut-brain axis: influence of microbiota on mood and mental health. Integr Med (Encinitas). 2018;17(4):28. Available from: https://pubmed.ncbi.nlm.nih.gov/31043907/
- Ferreira ACFM, Eveloff RJ, Freire M, Santos MTBR. The impact of oral-gut inflammation in cerebral palsy. Front Immunol. 2021;12:619262. Available from: https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2021.619262/full
- Loh JS, Mak WQ, Tan LKS, Ng CX, Chan HH, Yeow SH, Khaw KY, et al. Microbiota–gut–brain axis and its therapeutic applications in neurodegenerative diseases. Signal Transduct Target Ther. 2024;9(1):37. Available from: https://doi.org/10.1038/s41392-024-01743-1
- Pedrosa Carrasco AJ, Timmermann L, Pedrosa DJ. Management of constipation in patients with Parkinson’s disease. NPJ Parkinsons Dis. 2018;4(1):6. Available from: https://doi.org/10.1038/s41531-018-0042-8
- Becker A, Faßbender K, Oertel WH, Unger MM. A punch in the gut–intestinal inflammation links environmental factors to neurodegeneration in Parkinson's disease. Parkinsonism Relat Disord. 2019;60:43-45. Available from: https://doi.org/10.1016/j.parkreldis.2018.09.032
- Chuyue DY, Xu QJ, Chang RB. Vagal sensory neurons and gut-brain signaling. Curr Opin Neurobiol. 2020;62:133-140. Available from: https://doi.org/10.1016/j.conb.2020.03.006
- Forsythe P, Bienenstock J, Kunze WA. Vagal pathways for microbiome-brain-gut axis communication. In: Microbial endocrinology: the microbiota-gut-brain axis in health and disease. 2014. p. 115-133. Available from: https://doi.org/10.1007/978-1-4939-0897-4_5
- Farzi A, Fröhlich EE, Holzer P. Gut microbiota and the neuroendocrine system. Neurotherapeutics. 2018;15(1):5-22. Available from: https://doi.org/10.1007/s13311-017-0600-5
- Silva YP, Bernardi A, Frozza RL. The role of short-chain fatty acids from gut microbiota in gut-brain communication. Front Endocrinol (Lausanne). 2020;11:508738. Available from: https://doi.org/10.3389/fendo.2020.00025
- Fock E, Parnova R. Mechanisms of blood–brain barrier protection by microbiota-derived short-chain fatty acids. Cells. 2023;12(4):657. Available from: https://doi.org/10.3390/cells12040657
- Yang H, Kim D, Yang Y, Bagyinszky E, An SSA. TREM2 in neurodegenerative disorders: mutation spectrum, pathophysiology, and therapeutic targeting. Int J Mol Sci. 2025;26(15):7057. Available from: https://doi.org/10.3390/ijms26157057
- Biswas A, Petnicki-Ocwieja T, Kobayashi KS. Nod2: a key regulator linking microbiota to intestinal mucosal immunity. J Mol Med (Berl). 2012;90(1):15-24. Available from: https://doi.org/10.1007/s00109-011-0802-y
- Ramakrishna BS, Patankar R. Antibiotic-associated gut dysbiosis. J Assoc Physicians India. 2023;71(11):62-68. Available from: https://doi.org/10.59556/japi.71.0381
- Yang Z, Wei C, Li X, Yuan J, Gao X, Li B, Tang J, et al. Association between regular laxative use and incident dementia in UK Biobank participants. Neurology. 2023;100(16):e1702-e1711. Available from: https://doi.org/10.1212/wnl.0000000000207081
- Camacho M, Macleod AD, Maple-Grødem J, Evans JR, Breen DP, Cummins G, Williams-Gray CH, et al. Early constipation predicts faster dementia onset in Parkinson’s disease. NPJ Parkinsons Dis. 2021;7(1):45. Available from: https://doi.org/10.1038/s41531-021-00191-w
- Yao L, Liang W, Chen J, Wang Q, Huang X. Constipation in Parkinson’s disease: a systematic review and meta-analysis. Eur Neurol. 2023;86(1):34-44. Available from: https://doi.org/10.1159/000527513
- Warnecke T, Schäfer KH, Claus I, Del Tredici K, Jost WH. Gastrointestinal involvement in Parkinson’s disease: pathophysiology, diagnosis, and management. NPJ Parkinsons Dis. 2022;8(1):31. Available from: https://doi.org/10.1038/s41531-022-00295-x
- Kang J, Lee M, Park M, Lee J, Lee S, Park J, Kim T, et al. Slow gut transit increases the risk of Alzheimer’s disease: an integrated study of the bi-national cohort in South Korea and Japan and Alzheimer’s disease model mice. J Adv Res. 2024;65:283-295. Available from: https://doi.org/10.1016/j.jare.2023.12.010
- Rota L, Pellegrini C, Benvenuti L, Antonioli L, Fornai M, Blandizzi C, Colla E, et al. Constipation, deficit in colon contractions and alpha-synuclein inclusions within the colon precede motor abnormalities and neurodegeneration in the central nervous system in a mouse model of alpha-synucleinopathy. Transl Neurodegener. 2019;8(1):5. Available from: https://doi.org/10.1186/s40035-019-0146-z
- Kang SH, Lee J, Koh SB. Constipation is associated with mild cognitive impairment in patients with de novo Parkinson’s disease. J Mov Disord. 2021;15(1):38. Available from: https://doi.org/10.14802/jmd.21074
- Miyamoto T, Nakajima I, Arikawa T, Miyamoto M. Bowel movement frequency and difficult defecation using constipation assessment scale in patients with isolated REM sleep behavior disorder. Clin Parkinsonism Relat Disord. 2024;11:100269. Available from: https://doi.org/10.1016/j.prdoa.2024.100269
- Leta V, Urso D, Batzu L, Weintraub D, Titova N, Aarsland D, Chaudhuri KR, et al. Constipation is associated with development of cognitive impairment in de novo Parkinson’s disease: a longitudinal analysis of two international cohorts. J Parkinsons Dis. 2021;11(3):1209-1219. Available from: https://doi.org/10.3233/jpd-212570
- Su M, Wang S, Cheng O, Xie K, Peng J, Du X, Feng T, et al. Constipation is associated with emotional and cognitive impairment in patients with Parkinson’s disease: a clinical and brain functional study. Neuroscience. 2024;559:17-25. Available from: https://doi.org/10.1016/j.neuroscience.2024.08.027
- Wang Q, Yi T, Jiang X. Constipation and risk of cognitive impairment and dementia in adults: a systematic review and meta-analysis. Front Neurol. 2025;16:1600952. Available from: https://doi.org/10.3389/fneur.2025.1600952
- Soni S, Sasmal G, Bhardwaj V. Physiological concept of Vata Dosha in relation to brain function. J Ayurveda Integr Med Sci. 2024;9(5):169-171. Available from: https://jaims.in/jaims/article/view/3396
- Adams-Carr KL, Bestwick JP, Shribman S, Lees A, Schrag A, Noyce AJ. Constipation preceding Parkinson's disease: a systematic review and meta-analysis. J Neurol Neurosurg Psychiatry. 2016;87(7):710-716. Available from: https://doi.org/10.1136/jnnp-2015-311680
- Pfeiffer RF. Gastrointestinal dysfunction in Parkinson's disease. Lancet Neurol. 2003;2(2):107-116. Available from: https://doi.org/10.1016/s1474-4422(03)00307-7
- Gao H, He C, Hua R, Liang C, Wang B, Du Y, et al. Underlying beneficial effects of rhubarb on constipation-induced inflammation, disorder of gut microbiome and metabolism. Front Pharmacol. 2022;13:1048134. Available from: https://doi.org/10.3389/fphar.2022.1048134
- Jalanka J, Salonen A, Salojärvi J, Ritari J, Immonen O, Marciani L, et al. Effects of bowel cleansing on the intestinal microbiota. Gut. 2015;64(10):1562-1568. Available from: https://doi.org/10.1136/gutjnl-2014-307240
- Derrien M, van Hylckama Vlieg JE. Fate, activity, and impact of ingested bacteria within the human gut microbiota. Trends Microbiol. 2015;23(6):354-366. Available from: https://doi.org/10.1016/j.tim.2015.03.002
- O'Riordan KJ, Collins MK, Moloney GM, Knox EG, Aburto MR, Fülling C, et al. Short chain fatty acids: microbial metabolites for gut-brain axis signalling. Mol Cell Endocrinol. 2022;546:111572. Available from: https://doi.org/10.1016/j.mce.2022.111572
- Rajilic-Stojanovic M, Jonkers DM, Salonen A, Hanevik K, Raes J, Jalanka J, et al. Intestinal microbiota and diet in IBS: causes, consequences, or epiphenomena? Am J Gastroenterol. 2015;110(2):278-287. Available from: https://doi.org/10.1038/ajg.2014.427
- Nagata N, Takeuchi T, Masuoka H, Aoki R, Ishikane M, Iwamoto N, et al. Human gut microbiota and its metabolites impact immune responses in COVID-19 and its complications. Gastroenterology. 2023;164(2):272-288. Available from: https://doi.org/10.1053/j.gastro.2022.09.024
- Schirmer M, Smeekens SP, Vlamakis H, Jaeger M, Oosting M, Franzosa EA, et al. Linking the human gut microbiome to inflammatory cytokine production capacity. Cell. 2016;167(4):1125-1136. Available from: https://doi.org/10.1016/j.cell.2016.10.020
- Di Vincenzo F, Del Gaudio A, Pettit V, Lopetuso LR, Scaldaferri F. Gut microbiota, intestinal permeability, and systemic inflammation: a narrative review. Intern Emerg Med. 2024;19(2):275-293. Available from: https://doi.org/10.1007/s11739-023-03374-w
- Zhang Y, Zhu X, Yu X, Novák P, Gui Q, Yin K. Enhancing intestinal barrier efficiency: A novel metabolic diseases therapy. Front Nutr. 2023;10:1120168. Available from: https://doi.org/10.3389/fnut.2023.1120168
- Foster JA, Neufeld KAM. Gut–brain axis: how the microbiome influences anxiety and depression. Trends Neurosci. 2013;36(5):305-312. Available from: https://doi.org/10.1016/j.tins.2013.01.005
- Zimmermann M, Zimmermann-Kogadeeva M, Wegmann R, Goodman AL. Mapping human microbiome drug metabolism by gut bacteria and their genes. Nature. 2019;570(7762):462-467. Available from: https://doi.org/10.1038/s41586-019-1291-3
- Feng J, Zheng N, Fan X, Li S, Jiang Y, Yi X, et al. Association of laxatives use with incident dementia and modifying effect of genetic susceptibility: a population-based cohort study with propensity score matching. BMC Geriatr. 2023;23(1):122. Available from: https://doi.org/10.1186/s12877-023-03854-w
- Gordon M, MacDonald JK, Parker CE, Akobeng AK, Thomas AG. Osmotic and stimulant laxatives for the management of childhood constipation. Cochrane Database Syst Rev. 2016;(8). Available from: https://doi.org/10.1002/14651858.cd009118.pub3
- Koeth RA, Wang Z, Levison BS, Buffa JA, Org E, Sheehy BT, et al. Intestinal microbiota metabolism of L-carnitine and neuroinflammation. Nat Med. 2023;29(2):220-230. Available from: https://doi.org/10.1038/nm.3145
- Subbarayappa BV. Siddha medicine: an overview. Lancet. 1997;350(9094):1841-1844. Available from: https://doi.org/10.1016/s0140-6736(97)04223-2