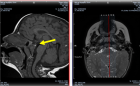
About Brown University
Brown University
Articles by Brown University
Parents Take-On Concussion: Advances in Sideline Research and Culture in Youth Sports
Published on: 16th March, 2017
OCLC Number/Unique Identifier: 7286354252
Identifying concussion and initiating removal from play is challenging for even the most diligent youth sports organizations. Empowering parents to implement removal from play protocols and sideline testing may be the most practical plan at community levels to protect young athletes. We developed paradigms for community-based youth sports teams that incorporated both standard concussion protocols and research investigations. The research studies were designed to determine how sideline tests of vision, cognition and balance augment the capacity for parents and other responsible adults to identify youth athletes with concussion in ice hockey, football, lacrosse and cheerleading. Research-based sideline tests were performed at pre-season baseline sessions and during the season at the time of injury or as soon as symptoms were recognized by trained volunteer parent team testers. The combination of standard concussion protocols and research studies were performed for 510 athletes, aged 5-17 years, over 2.5 years through 5 athletic seasons. To implement the protocols and studies, approximately 80 student volunteers and parents were educated and trained on early concussion recognition and on baseline and sideline test administration. Over 80% of parent-identified head injuries were physician-confirmed concussions. Of the sideline tests performed, over two-thirds were administered within 24 hours of injury; the rest were performed within an average of 2.6 days post-injury since some athletes had delayed development of symptoms. Removal from play guidelines and standard concussion evaluation protocols were maintained in the context of the sideline testing research investigations. Based on this observational study, parents of youth athletes can be successfully empowered to perform rapid sideline tests in the context of existing concussion protocols. Implementation of objective testing may improve concussion identification and shift the culture of advocacy and responsibility towards parent groups to promote safety of young athletes. Ongoing investigations will further examine the impact of these programs on concussion management in youth sports.
Low sensitivity of the careHPV™ Assay for detection of Oncogenic Human Papillomavirus in cervical samples from HIV-infected and HIV-uninfected Kenyan women
Published on: 30th January, 2020
OCLC Number/Unique Identifier: 8535181241
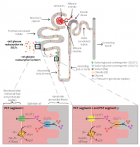
Background: Human papillomavirus (HPV) infection causes cervical cancer (CC), a common malignancy among Kenyan women. New CC screening methods rely on oncogenic HPV (“high-risk”, or HR-HPV) detection, but most have not been evaluated in swabs from Kenyan women.
Methods: HPV typing was performed on 155 cervical swabs from Kenyan women using the Roche Linear Array® (LA) and careHPV™ (careHPV) assays. Detection of 14 oncogenic HPV types in careHPV assay was compared to LA results.
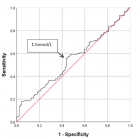
Results: Compared to LA, sensitivity and specificity of careHPV assay was 53.0% and 80.9%, respectively. The sensitivity and specificity of careHPV in swabs from women with cervical dysplasia was 74.1% and 65.2%, respectively. The sensitivity and specificity of careHPV in swabs from HIV-infected women was 55.9% and of 96.4%, respectively. Overall agreements of careHPV assay with LA was substantial.
Conclusion: The results for careHPV assay are promising for oncogenic HPV detection in Kenyan women. The low sensitivity of careHPV for detection of HR-HPV could limit it’s benefit as a screening tool. Thus, a full clinical validation study is highly desirable before the careHPV assay can be accepted for cervical cancer screening.
Impact of mandibular advancement device in quantitative electroencephalogram and sleep quality in mild to severe obstructive sleep apnea
Published on: 30th December, 2020
OCLC Number/Unique Identifier: 8899350400
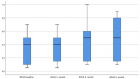
Sleep related breathing disorders (SRBD) are among seven well-established major categories of sleep disorders defined in the third edition of The International Classification of Sleep Disorders (ICSD-3), and Obstructive Sleep Apnea (OSA) is the most common SRBD [1,2]. Several studies have demonstrated that obstructive sleep apnea treatment increases the quality of life in OSA patients [3-8]. Indeed, excessive daytime sleepiness (EDS), cognitive impairment (e.g., deficits in attention-concentration, memory, dexterity, and creativity), traffic accidents, and deterioration of social activities are frequently reported in untreated patients [9-11]. Furthermore, an increase in cardiovascular morbidities and mortality (systemic hypertension, stroke, cardiac arrhythmias, pulmonary arterial hypertension, heart failure) [12], metabolic dysfunction, cerebrovascular ischemic events and chemical/structural central nervous system cellular injuries (gray/white matter) has been reported in OSA patients [13-17].
Continuous positive airway pressure (CPAP) therapy is considered the gold standard for treatment of moderate-severe OSA, nevertheless there is an increasing body of evidence supporting the usefulness of mandibular advancement devices (MADs) for improving quality of life and respiratory parameters even among patients with a high severity of OSA burden [5,10,18,19]. According to the standard of care of the American Academy of Sleep Medicine (AASM), MADs are indicated for mild to moderate OSA particularly in the context of CPAP intolerance or refusal, surgical contraindication, or the need for a short-term substitute therapy [9,15,20-22]. In Cuba, CPAP machines are not readily available; they are expensive and the majority of OSA patients cannot obtain this mode of therapy. Taking into account this problem, our hypothesis was based in the scientific evidences of MAD effectiveness, considering that low cost MADs could offer a reasonable alternative treatment for patients with OSA where CPAP technology are not handy. In this way our purpose was to assess the efficacy of one of the most simple, low cost, manufactured monoblock MAD models (SAS de Zúrich) in terms of improvements in cerebral function, sleep quality and drowsiness reports in a group of Cuban OSA patients with mild to severe disease. Outcome measures included changes in the brain electrical activity, sleep quality, and respiratory parameters, measured by EEG recording with qEEG analysis and polysomnographic studies correspondingly, which were recorded before and during treatment with an MAD, as well as subjective/objective improvements in daytime alertness.
Patient, disease and surgeon predictors of successful bilateral sentinel lymph node mapping for endometrial cancer: A retrospective, multicenter analysis
Published on: 14th July, 2022
Objective: Sentinel lymph node mapping is an acceptable standard for lymph node evaluation in patients with endometrial cancer. The purpose of this study was to evaluate the adoption of this technique at two academic institutions, including which patient and disease features are associated with rates of successfully identifying sentinel lymph nodes with fluorescent mapping. In addition, we sought to characterize if and how surgeons experience the technique related to successful bilateral sentinel lymph node mapping. Methods: A retrospective chart review was performed of patients at two academic institutions who underwent sentinel lymph node mapping prior to a minimally invasive hysterectomy for endometrial cancer over the first 30 months during which the technique was adopted at each institution. A modified Poisson regression model was used to determine the relationships between patient, disease, and surgeon factors on outcomes of sentinel lymph node mapping. Results: A total of 460 charts were reviewed. The mean age was 64 and the median body mass index was 34.2. The most disease was stage I (83%), endometrioid (89%), and Grade I (64%). The bilateral sentinel lymph node mapping success rate was 65%, while unilateral or bilateral success occurred in 91% of cases. Sentinel lymph node mapping was significantly more likely to be successful in premenopausal women (RR 1.25; 95% CI 1.07 - 1.46; p = 0.005) and Asian women (RR 1.48; 95% CI 1.3-1.68; p < 0.001). BMI was not significantly predictive of mapping success (RR 1.03; 95% CI 1.00 - 1.07; p = 0.05). Increasing surgeon experience with the technique did predict successful bilateral sentinel lymph node mapping (RR 1.02; 95% CI 1.00 - 1.03; p = 0.02).Conclusion: Premenopausal status and surgeon experience with the technique increases the likelihood of bilateral sentinel lymph node detection for endometrial cancer.

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."